read time: 4 min
Health & Wellness
Understanding cell regeneration
“You’re a new person every 7 years!”
The claim that our cells change every 7 years is a factoid you may have heard at parties, but is it actually true?
Your body holds a lot of cells. In fact, the human body is composed of about 37 trillion cells.1 Do every single 1 of those 37 trillion cells change every 7 years? And why do our cells need to change, anyway? Let’s dive into why this process, called cell regeneration, is so important for your health.
What is cell regeneration?
Cell regeneration is probably happening in your body right now. And that’s a good thing! In many species, regeneration plays a big role in helping the body heal itself and remain healthy. It does this by replacing or restoring damaged cells, tissues, and organs.
Here’s how it works. When you’re injured or sick, your cells are damanged. Your cells also die all the time, a natural process called apopstosis. Your body needs to replace damaged or dead cells with new, healthy ones. To create these new cells, the existing healthy cells in your body go through cell division, or mitosis. During mitosis, a cell splits into 2 cells, each with an identical set of chromosomes. These new healthy cells can then be used to help you heal.
Some creatures, like starfish and salamanders, can use this process to regenerate entire limbs. While the human body isn’t quite that talented, regeneration in our cells, tissues, and organs is happening all the time. Think of regeneration as providing general maintenance for your body, constantly taking stock of your health and repairing things whenever possible.
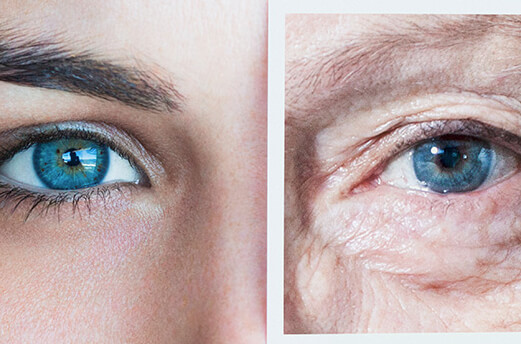
So, let’s get back to that “7 year” talking point. Do all of the cells in your body really regenerate that often? The answer is yes… and also no. While it’s true that your cells regenerate on average every 7-10 years,2 there’s a lot of variation. Your skin cells, for example, are replaced every few weeks.3 In fact, you lose close to 500 million skin cells every day.4 Cells in your skeletal muscles, on the other hand, take as long as 15 years to regenerate.2
What cells in your body are never replaced?
Some of your cells aren’t able to regenerate at all. Spinal cord injuries typically lead to damaged nerves that aren’t able to be repaired. However, scientists are actively working on ways to promote regeneration within spinal cord nerves.5
It used to be accepted that adult brain cells weren’t able to heal themselves. More recent findings, however, have shown encouraging signs that brain cells may be capable of regrowth. A 2020 study found that after an adult experiences a brain injury, cells revert to a less mature state. From there, they may be able to regrow.6 The more scientists learn about the cell regeneration process, the better equipped they are to explore new ways to help the body heal.
Even if we don’t sustain large injuries, or injure a part of our body that can’t go through regeneration, our bodies can’t remain healthy forever. As we age, our cells stop dividing as often. So, as we get older and our cells start dying, there may not be enough new cells created to replace the dead ones. This makes it more difficult for our bodies to heal properly.
What happens when cell regeneration goes wrong?
We depend on cell regeneration to heal our bodies. Mitosis, however, can leave room for mistakes while cells are dividing. These mistakes can lead to the creation of cancerous cells.
Healthy cells only divide when it’s necessary (when you’re injured, for example) and the process stops when your body gets enough new cells. Cancer cells, however, don’t listen when your body signals that it’s time for them to stop growing. They continue to get bigger and divide. These overgrown cells can then form tumors. In blood cancers, these build up an army of abnormal cells in your blood.
Stem cell research: the future of regenerative medicine
We know that some of our cells can regenerate while others can’t. We also know that cancerous cells can’t undergo regeneration to be replaced with healthy ones. But what if we could use cells that can regenerate to help repair the parts of our body that can’t heal on their own?
Enter stem cell research. Stem cells have the ability to develop into different types of cells. Most cells are specialized to 1 part of the body (heart cells or brain cells, for example). Stem cells, however, aren’t stuck committing to being just 1 type of cell. This ability gives them the potential to repair or replace other types of damaged cells.
While it’s hard to say exactly how many stem cells we have in our body, a 2018 study found that, at any given time, we have between 50,000 and 200,000 stem cells actively making white blood cells.7 And that’s just in 1 area of our body!
Stem cells are the heart of a field called regenerative medicine, which is dedicated to finding new ways to repair or replace damaged cells, tissues, and organs. Scientists hope that some day soon regenerative medicine can be used to treat illnesses like heart disease, cancer, and Alzheimer's disease.8
Cell regeneration doesn’t just keep your body healthy today—it may also be the key to a healthier future for millions with hard-to-treat diseases.
References
1 Roy AL, Conroy RS. Toward mapping the human body at a cellular resolution. Mol Biol Cell. 2018 Aug 1;29(15):1779-1785. doi: 10.1091/mbc.E18-04-0260. PMID: 30058989; PMCID: PMC6085824
2 Spalding KL, Bhardwaj RD, Buchholz BA, et al. Retrospective birth dating of cells in humans. Cell. 2005 July 15; 1(122):133-143. doi: 10.1016/j.cell.2005.04.028
3 Weschler CJ, Langer S, Fischer A, et al. Squalene and cholesterol in dust from Danish homes and daycare centers. Environmental Science and Technology. 2011; 45(9): 3872-3879. doi: 10.1021/es103894r
4 Milstone, LM. Epiderman desquamation. Journal of Dermatological Science. 2004 Dec 1; 3(36): 131-140. doi: 10.1016/j.jdermsci.2004.05.004
5 Boosting energy levels within damaged nerves may help them heal.
National Institutes of Health (NIH). March 3, 2020. Accessed September 29, 2022. https://www.nih.gov/news-events/news-releases/boosting-energy-levels-within-damaged-nerves-may-help-them-heal
6 Poplawski GHD, Kawaguchi R, Van Niekerk E, et al. Injured adult neurons regress to an embryonic transcriptional growth state. Nature. 2020 April 15; 581: 77–82. doi: 10.1038/s41586-020-2200-5
7 Lee-Six H, Øbro NF, Shepherd MS, et al. Population dynamics of normal human blood inferred from somatic mutations. Nature. 2018 September 5; 561: 473–478. doi: 1038/s41586-018-0497-0
8 Regenerative medicine innovation project.
National Institutes of Health (NIH). N.d. Accessed September 29, 2022. https://www.nih.gov/rmi